- Visibility 235 Views
- Downloads 53 Downloads
- Permissions
- DOI 10.18231/j.ijohd.2024.039
-
CrossMark
- Citation
A comparative evaluation of cytotoxicity and corrosion resistance between titanium and stainless steel orthodontic mini implants: An in vitro study
Abstract
Introduction: Micro-implants made from metals, have been developed and modified to apply to many of the orthodontic situations one of which is absolute anchorage. The continuous use of metals in oral cavity has led to a great number of laboratory and clinical studies on the damaging effects of this products to person’s general health. Owing to this, it is must to rule out the best mini-implant with least cytotoxicity and high corrosion resistance.
Aim and Objective: The aim of this in-vitro study was to evaluate and compare the cytotoxicity and corrosion resistance of Titanium and Stainless steel Orthodontic mini-implants.
Materials and Methods: The sample comprised of 20 mini-implants which were divided into 4 groups, each group consisted of 5 mini-implants.2 groups of Titanium mini-implant and 2 groups of Stainless Steel mini-implant. Subsequently, each sample was submerged in artificial saliva for 60 and 90 days, at 37°C. After this, saliva with the mini-implant corrosive product extracts were evaluated for cytotoxicity and the mini-implants were tested for corrosion resistance.
Result: In intergroup comparison for cytotoxicity, significant values seen with Orlus mini-implants and least significant values with Favanchor mini-implants. Whereas, for corrosion resistance, a highly significant difference seen for values between all the 4 mini-implants with higher values in Favanchor mini-implants.
Conclusion: There was a highly significant difference for all the 4 mini-implants, with least cytotoxicity seen in Titanium mini-implants than Stainless Steel mini-implants. Mini-implants of all the 4 groups exhibited good corrosion resistance, with comparatively high corrosion resistance seen in Titanium mini-implants than Stainless Steel mini-implants.
Introduction
Orthodontic treatment is a complex procedure, requiring a balance with the orthodontist biomechanics for an individual patient. Anchorage control is considered to be a keystone of the orthodontic force system. Anchorage which is offered by the teeth that resist the forces of reaction generated by the active components of the appliance.
Until now, various techniques to reinforce anchorage have been discovered and used in orthodontic practice. When anchorage demand is maximum additional aids are often needed to support the anchoring teeth. Today, the much-desired absolute anchorage is provided by mini implants and more significantly, the use of these anchorage modalities does not rely on patient's compliance.[1]
Microimplants made from metals, which were originally used only for surgical plates, have been developed and modified to apply to many of the orthodontic purpose. Its use has been applied to multiple orthodontic situations, such as for retraction, intrusion, extrusion, distalisation, mesialisation, and so on. Most importantly, with their small size, they can be placed in the alveolar bone of adjacent teeth without disrupting adjacent structures.[1], [2], [3], [4], [5], [6], [7], [8], [9], [10]
The continuous use of metals in oral cavity has led to a great number of laboratory and clinical studies on the damaging effects of corrosion products to person’s general health. Also, the oral cavity is considered to have a corrosive environment. So, the corrosion resistance of orthodontic metals depends on the oral environment which is influenced by various variables, such as the quantity and quality of saliva, pH of food and brews, etc. and hence the release of metal ions from orthodontic metals is of great concern.[1]
In Orthodontics, the majorly used alloys for mini-implants are Titanium alloys and Stainless Steel alloys. Both of these alloys are said to be corroded to a certain extent in the acidic oral environment. Owing to this, it is must to rule out the cytotoxicity and corrosion resistance of orthodontic mini-implants, which will help us to determine the best mini-implant with least cytotoxicity and high corrosion resistance to be used in oral cavity.[1]
Although in vitro studies do not replicate the same oral environment, standard assays are useful to assess the cytotoxicity and biocompatibility of temporary anchorage devices, which are mini-implants. Hence, the aim of this in vitro study was to evaluate and compare the cytotoxicity and corrosion resistance of Titanium and Stainless steel mini-implants for orthodontic anchorage.
Materials and Methods
The sample comprised of 20 orthodontic mini-implants which were further divided into 4 groups, each group consisted of 5 orthodontic mini-implants.
Study material include
4 Groups of Orthodontic mini implants were evaluated:
Two groups of Titanium mini implant([Figure 1])
Group 1 = Vector Task mini-implants
Group 2 = Orlus mini-implants
Two groups of Stainless Steel mini implant ([Figure 1])
Group 3 = Bio ray mini-implants
Group 4 = Favanchor mini-implants

Methodology
Each orthodontic mini-implants was autoclaved at 120°C for 30 minutes. Subsequently, each sample was submerged in artificial saliva for 60 and 90 days, at 37°C. Tubes containing only artificial saliva, without mini-implants, were stored as negative control. After this, mini-implants were removed, washed, dried and stored in new sterile airtight plastic tubes, and saliva with the mini-implant corrosive product extracts were then evaluated for cytotoxicity and the mini-implants were tested for corrosion resistance.
L929 cell culture formation
The cell line used for this study was L929 cells procured from NCCs Pune, India. The cell line was maintained in 96 wells micro titer plate containing DMEM media supplemented with 10% heat inactivated fetal calf serum (FCS), containing 5% of mixture of Gentamicin (10ug), Penicillin (100 Units/ ml) and Streptomycin (100μg/ml) in presence of 5% CO2 at 37ºC for 48-72 hours. Artificial saliva was used as negative control, since it is not cytotoxic to cell-culture. A cytokine capable of destroying L929 cells, which is the tumor necrosis factor (TNF), after approximately 20 hours of culture, was used as positive control.
Cytotoxicity assays
Aliquots of 100μL of L929 cell suspension were pipetted into 96 flat bottom plates and were incubated at 37o C for 48 hours. TNF solution of 100μL aliquots was placed in each well containing 100μL of L929 cells.
Determination of cell viability with the help of crystal violet (CV) colorimetric assay
Cells were seeded into 96 well microtiter plate at a concentration of 2x105 cells per well and incubated overnight in standard media, so as to facilitate the attachment of the cells.
Next day the media was removed and the mini- implants extracts was added to the wells at a concentration of 20% and incubated for 48 hours.
After 48 hours, the media was removed, the wells were washed with PBS and stained with crystal violet for 30 minutes. Extra stain was removed by washing with distilled water.
For solubilisation of bound crystal violet, 33% glacial acetic acid was added. Absorbance was measured by a spectrophotometer at 570nm. The resulting stained solution, were then evaluated for total number of viable cells retained on the plates. Culture medium without cells were blank.
Determination of cellular metabolism with the help of MTT colorimetric assay
In vitro growth inhibition effect of test compound was assessed by colorimetric or spectrophotometric determination of conversion of MTT into “Formazan blue” by living cells. The supernatant from the plate were removed and fresh DMEM solution were added and treated with the artificial saliva treated with the implants to respective wells.([Figure 2])
After 48hrs incubation at 37ºC in a humidified atmosphere of 5% CO2, stock solution of MTT was added to each well (20μl, 5mg per ml in sterile PBS) and further incubated for 4 hrs. After four hours, the supernatant was carefully aspirated. The precipitated crystals of “Formazan blue’ were solubilized by adding DMSO (100μl) and optical density was measured at wavelength of 595nm by using LISA plus. ([Figure 3])
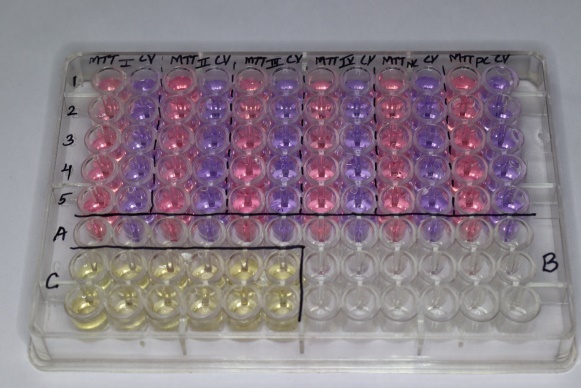
Corrosion resistance
Determination of corrosion resistance by Field emission scanning electron microscopy (FESEM)
In order to assess mini-implants surface characteristics, a sample of each artificial saliva immersion group and a sterile packaged control sample were taken randomly and examined with the help of field emission scanning electron microscope (FESEM) to determine surface characteristics. Surface topography of the mini-implants were examined under magnification.
Determination of corrosion products by inductively coupled plasma – optical emission spectrometry (ICP-OES)
Mini-implant extract solutions obtained after 60 and 90-days immersion period in artificial saliva were studied with the help of ICP-OES machine, to conclude and quantify the amount of aluminium and vanadium ions released due to corrosion and oxidation.([Figure 4])

Statistical procedures
All data were entered into a computer by giving coding system, proofed for entry errors, data obtained was compiled on a MS Office Excel Sheet (version 2019, Microsoft Redmond Campus, Redmond, Washington, United States). Data was subjected to statistical analysis using statistical package for social sciences (SPSS v 26.0, IBM). Descriptive statistics like frequencies and percentage for categorical data, Mean & SD for numerical data has been depicted. Normality of numerical data was checked using Shapiro-Wilk test & was found that the data followed a normal curve; hence parametric tests have been used for comparisons. Inter group comparison (>2 groups) was done using one way ANOVA followed by pair wise comparison using post hoc test. For all the statistical tests, p<0.05 was considered to be statistically significant, keeping α error at 5% and β error at 20%, thus giving a power to the study as 80%.
Results
Cytotoxicity
In Intergroup comparison, there was a highly significant difference seen for all the 4 groups of mini-implants, With more significant values seen with Group 2, i.e. Titanium - Orlus implants and least significant values with Group 4, i.e. Stainless Steel - Favanchor implants. There was a statistically highly significant difference seen for the values between all the pairs of groups (p<0.01, 0.05) except for MTT at 90 days between Vector Task and Orlus, and between Vector Task and Bioray, where there was a statistically non-significant difference seen (p>0.05).
In the Intra-group comparison, there was a statistically significant difference seen between the time interval of 60 and 90 days in the 2 groups of orthodontic mini-implants, i.e. between Orlus and Bioray, except for the Intra-group comparison of MTT for Vector task mini-implants and Favanchor mini-implants, where there was a non-significant difference seen.
Corrosion resistance
For FESEM, there was a statistically non-significant difference seen for the values between all the 4 mini-implants (p>0.05) at 60 days but at 90 day, there was a statistically highly significant difference seen for the values between all the 4 mini-implants (p<0.01), with higher values in Favanchor mini-implants. For ICP-OES, the concentrations of Aluminium and Vanadium metal ions release were below the limit of quantification.


In [Figure 5] depicts inter-group comparison of MTT assay, at 60 day and 90 days, between all the 4 groups of mini-implants, which were statistically significant.
In [Figure 6] depicts intra-group comparison of MTT assay, between 60 day and 90 days, between all the 4 groups of mini-implants, wherein Group2 - Orlus and Group 3 -Bioray mini-implant which were statistically significant and Group 1 – Vector Task and Group 4 – Favanchor mini-implants were non-significant.


In [Figure 7] depicts inter-group comparison of CV test, at 60 day and 90 days, between all the 4 groups of mini-implants, which were statistically significant.
[Figure 8] depicts intra-group comparison of CV test, between 60 day and 90 days, between all the 4 groups of mini-implants, which were statistically significant between that time intervals.


[Figure 9] depicts inter group comparison of SEM data, where a statistically non-significant difference was seen for all the groups of mini-implants at 60 days, but there was a statistically highly significant difference seen for all the groups of mini-implants at 90 days, with higher values seen in group 4, i.e. Favanchor mini-implant.
[Figure 10] depicts intra group comparison of SEM data, where a statistically significant difference was seen for the values between the time intervals (60 day to 90 days) for bioray mini-implant, but a statistically non-significant difference was seen for the rest three mini-implants group.
Discussion
In this study, orthodontic mini-implants ready for clinical use as anchorage devices were evaluated for their potential side effect.
Mini-implants are an effective and very well tolerated appliance for skeletal anchorage, and have become the gold standard for orthodontic biomechanics for anchorage in adults. They are available in a variety of lengths, shapes, diameters, and different alloy compositions.[11]
These implants are manufactured almost extensively from Stainless Steel and Titanium alloy (Ti6Al4V) with the addition of certain metals mainly aluminium and vanadium ions for greater strength and fatigue resistance to withstand orthodontic forces for any kind of tooth movement. However, aluminium and vanadium have been associated with certain amount of side effects to the human body.[12], [13]
These implant usually contain at least 4 metals, or often 6 or 8 metals, including titanium, chromium, vanadium, nickel, aluminium, iron, etc. Moreover, this alloy can lead to corrosion of orthodontic mini-implants because of the low corrosion resistance of the titanium alloy in the body fluids.[10]
Materials used in the oral cavity must be nontoxic as well as biocompatible, have good mechanical properties, and be able to have corrosion resistance. The most common alloys and metals used in dentistry can be revealed to corrosion in vivo that makes them cytotoxic.[10]
Biosafety and biocompatibility are of major concern to the clinical application of dental materials including mini-implants for TAD’s. In vitro cytotoxicity tests are given by ISO to determine acute cytotoxicity of a material (International Standard Organization) and also help in better knowing the pathogenicity of sub-acute effects.[14]
The cytotoxicity in our study was determined by using L929 cell line, with the help of CV test and MTT assay.
In this test, crystal violet (CV) dye binds to proteins and DNA of viable cells, and thus, the attached cells are stained with CV dye. Cells lose their adherence during cell death and are then lost from the population of cells, thus reducing the amount of crystal violet staining in the culture. However, Crystal violet assay is insensitive to changes in cellular metabolic activity and to measure cell proliferation rate.[15]
Therefore for better conformation of results, we have examined our samples through MTT assay, which measures viable cells in relatively high throughput (96-well plates) without the need for elaborate counting of cells. Hence, it is most commonly used to determine cytotoxicity of several drugs at different cellular concentrations.[16]
MTT is considered to be safe, easy to use, has got a high reproducibility, and is widely used to determine both viability of cells and also cytotoxicity tests.[15] In the present study, for determining cytotoxicity, we evaluated all the 4 groups of orthodontic mini-implants with the help of MTT and CV colorimetric assays and results were such that all the groups showed highly significant values, with more significant in group 2, i.e. Orlus mini-implant, being the best amongst all and the least significant with group 4, i.e. Favanchor mini-implants. Hence, here we can state that, none of the evaluated mini-implants had an adverse effects on the cell viability, but a significant reduction in the number of cells were seen with Stainless Steel mini-implants.
TADs have shown to cause fluctuating cellular reactions on various cell types in the previous studies. In an in vitro study, Malkoc et al. noticed that the Ti-6Al-4V alloy in Vector TAS (ORMCO) and in MTN (Turkey) mini-implant, significantly reduced the MC3T3-E1 (mouse osteoblasts) cell viability at 190 hr. in contrast to IMTEC Ortho (3M Unitek, Europe) and Abso Anchor (Dentos, Incorporated, Dong-In-Dong Jung-Gu Daugu, South Korea) mini-implant. And reported that none of the evaluated TADs had significant adverse effects on human gingival fibroblasts.[14]
Our results are consistent with those reported by Malkoc et al. where they evaluated cytotoxicity in 5 types of mini-implants majorly made of 2 alloys, i.e. Stainless steel and Titanium, using a real-time cell analysis and they concluded that no adverse effects were seen on the gingival fibroblasts in either of the material, but a significant reduction in the number of osteoblasts viability was seen for stainless steel mini-implants, which stated Titanium alloys are better than Stainless Steel alloys.[10]
Thus the result of our study is in close agreement with the above studies, where titanium and stainless steel mini-implants exhibited almost similar cytotoxicity, with comparatively less cytotoxicity seen with Titanium mini-implants than Stainless Steel mini-implants.
Moving ahead to corrosion resistance, Yu J et al, suggested that pitting is the most likely causative factor to initiate corrosion fatigue, pitting corrosion be the primary cause of deterioration of corrosion fatigue resistance for stainless steel in human body fluids and the effective modality to reduce the risk of corrosion fatigue for stainless steels implants is to improve their pitting corrosion resistance.[17]
Recent studies have stated that even though titanium alloys are suggested to be highly corrosion-resistant because of the stable passive titanium oxide layer on its surface, they are not inert to corrosion attack. Retrieved mini-implants showed significant surface and structural alterations, such as corrosion, dullness and blunting of tips and threads. Their implant surfaces showed interactions and adsorption of several elements, such as calcium, at their body region.[18]
In our study, we evaluated the mini-implants for 60 and 90 days, taking into consideration that the mini-implants stays in the oral cavity for more than 2 months for greater effectiveness.
Gurappa et al. in one of his study mentioned that the maximum breakdown potential was seen for the titanium alloy, Ti – 6Al – 4V, and followed by the cobalt alloy and the minimum was observed with stainless steel. The maximum corrosion rate was seen with stainless steel followed by the cobalt alloy. The minimum corrosion rate was observed with titanium alloy, Ti – 6Al – 4V. Furthermore, they added, Ti – 6Al – 4V alloy exhibits the minimum corrosion rate and maximum breakdown potential when compared to other materials. They also stated that any metal/alloy supposed to be used as a biomaterial should have excellent pitting and crevice corrosion resistance and concluded that titanium and cobalt alloys have excellent pitting and crevice corrosion resistance, while stainless steel is highly susceptible to pitting and crevice corrosion. From their study, they have stated that the ability of stainless steel to repassivate is considerably less compared to other materials used.[19]
Their result justifies with the result of our study, where we evaluated both the Titanium and Stainless Steel orthodontic mini-implants with the help Field Emission Scanning Electron Microscope (FESEM) and inductively coupled plasma – optical emission spectrometry (ICP-OES).
We have tested our samples with the help of FESEM as it is considered to be the only electron source which is designed for high-resolution of imaging and is suitable for various kinds of materials in field emission, which utilizes (FEG) Field emitter gun to emit electrons. FESEM is based on a technology of high-resolution of images and different contrasting methods which aims for a comprehensive characterization of specimens. High-quality images and low-voltage with slight electrical charging (accelerating voltages ranging from 0.5 to 30 kV) of samples can been obtained using FESEM testing machines.[20]
Hence, we conducted our study with the help of FESEM in order to achieve better and accurate results. So according to FESEM testing results, there was a statistically non-significant difference in all the 4 orthodontic mini-implants group at 60 days, but there was a statistically significant difference in all the 4 orthodontic mini-implants group at 90 days, with the higher values of corrosion seen in group 4, i.e Stainless Steel, Favanchor group, which suggests maximum corrosion was seen with Stainless Steel. However, all orthodontic mini-implants immersed for 90 days showed darkened spots and more adhered particles which suggests decreased corrosion resistance.
Taking into consideration that 90 days was the maximum period that the mini-implants were subjected to artificial saliva, a time in which all samples remained stationary, not addressed to any orthodontic force in which the results suggested no signs of corrosion in the 4 groups of orthodontic mini-implants, the presence of manufacturing/corrosion defects on the Favanchor mini-implants surface causes concern.
Therefore we can state that, both the Titanium and Stainless Steel mini-implants can be used effectively when orthodontic anchorage is considered, but Titanium remains at a little higher version when compared to Stainless Steel.
Conclusion
Cytotoxicity
Less cytotoxicity was observed in L929 cell morphological evaluation, cell damage, growth inhibition, and alteration of cellular metabolism. There was a highly significant difference for all the 4 mini-implants, with least cytotoxicity seen in Titanium mini-implants than Stainless Steel mini-implants. In Titanium - Orlus mini-implant showed less cytotoxicity followed by Vector Task mini-implant. In Stainless Steel - Bioray mini-implant showed less cytotoxicity followed by Favanchor mini-implant.
Corrosion resistance
Mini-implants of all the 4 groups exhibited good corrosion resistance, with comparatively high corrosion resistance seen in Titanium mini-implants than Stainless Steel mini-implants. In Titanium – Vector Task implant showed comparatively high corrosion resistance than Orlus mini-implants. In Stainless steel - Bioray implant showed high corrosion resistance followed by Favanchor mini-implants. There were no release of Aluminium and Vanadium ions in all the 4 groups of extracted solutions.
Hence, we can conclude that, Titanium and Stainless Steel mini-implants exhibited almost similar amount of cytotoxicity and corrosion resistance, with better values seen in Titanium mini-implants and both can be used when orthodontic anchorage is considered.
Conflict of Interest
None.
Source of Funding
None.
Ethical Approval
Study is approved by ethical committee of institute and MUHS research committee.
Acknowledgement
I would like to express my sincere gratitude to my mentor and guide Dr. Ajit Kalia (HOD and Professor, department of orthodontics, M.A Rangoonwala College of Dental Science and Research Centre, Pune) who provided inspiration and guidance behind the idea of the study.
References
- Alves C, Segurado M, Dorta M, Dias F, Lenza M, Lenza M. Evaluation of cytotoxicity and corrosion resistance of orthodontic mini-implants. Dental Press J Orthod. 2016;21(5):39-6. [Google Scholar]
- Chen C, Chang C, Hsieh C, Tseng Y, Shen Y, Huang I. The use of microimplants in orthodontic anchorage. J Oral Maxillofac Surg. 2006;64(8):1209-13. [Google Scholar]
- Eliades T, Bourauel C. Intraoral aging of orthodontic materials: the picture we miss and its clinical relevance. Am J Orthod Dentofacial Orthop. 2005;127(4):403-12. [Google Scholar]
- Ashith M, Shetty B, Shekatkar Y, Mangal U, Mithun K. Assessment of immediate loading with mini-implant anchorage in critical anchorage cases between stainless steel versus titanium miniscrew implants: a controlled clinical trial. Biomed Pharmacol J. 2018;11(2):971-7. [Google Scholar]
- Disegi J, Eschbach L. Stainless steel in bone surgery. Injury. 2000;31(4):2-6. [Google Scholar]
- Siva S, Sivakumar A, Subramanian A, Dinesh S. Cytotoxicity of SK surgical implants used for anchorage in orthodontics: In vitro study. Drug Invention Today. 2018;10(1):2927-9. [Google Scholar]
- Morais L, Serra G, Muller C, Andrade L, Palermo E, Elias C. Titanium alloy mini-implants for orthodontic anchorage: immediate loading and metal ion release. Acta Biomater. 2007;3(3):331-9. [Google Scholar]
- Brown R, Sexton B, Chu T, Katona T, Stewart K, Kyung H. Comparison of stainless steel and titanium alloy orthodontic miniscrew implants: a mechanical and histologic analysis. Am J Orthod Dentofacial Orthop. 2014;145(4):496-504. [Google Scholar]
- Pithon M, Santos R, Martins F, Medeiros P, Romanos M. Citotoxicity of orthodontic mini-implants. Rev Clín Pesq Odontol Curitiba. 2010;6(2):141-6. [Google Scholar]
- Malkoç S, Öztürk F, Çörekçi B, Bozkurt B, Hakki S. Real-time cell analysis of the cytotoxicity of orthodontic mini-implants on human gingival fibroblasts and mouse osteoblasts. Am J Orthod Dentofacial Orthop. 2012;141(4):419-26. [Google Scholar]
- Burmann P, Ruschel H, Vargas I, Verney JD, Kramer P. Titanium alloy orthodontic mini-implants: scanning electron microscopic and metallographic analyses.. Acta Odontol Latinoam. 2015;28(1):42-7. [Google Scholar]
- Morais LD, Serra G, Palermo E, Andrade L, Müller C, Meyers M. Systemic levels of metallic ions released from orthodontic mini-implants. Am J Orthod Dentofacial Orthop. 2009;135(4):522-9. [Google Scholar]
- Ananthanarayanan V, Padmanabhan S, Chitharanjan A. A comparative evaluation of ion release from different commercially-available orthodontic mini-implants-an in-vitro study. Aust Orthod J. 2016;32(2):165-74. [Google Scholar]
- Chen Z, Patwari M, Liu D. Cytotoxicity of orthodontic temporary anchorage devices on human periodontal ligament fibroblasts in vitro. Clin Exp Dent Res. 2019;5(6):648-54. [Google Scholar]
- Flick D, Gifford G. Comparison of in vitro cell cytotoxic assays for tumor necrosis factor. J Immunol Methods. 1984;68(1-2):167-75. [Google Scholar]
- Gerlier D, Thomasset N. Use of MTT colorimetric assay to measure cell activation. J Immunol Methods. 1986;94(1-2):57-63. [Google Scholar]
- Yu J, Zhao Z, Li L. Corrosion fatigue resistances of surgical implant stainless steels and titanium alloy.. Corrosion sci. 1993;35(1-4):587-97. [Google Scholar]
- Leung V, Darvell B. Artificial salivas for in vitro studies of dental materials. J dent. 1997;25(6):475-84. [Google Scholar]
- Gurappa I. Characterization of different materials for corrosion resistance under simulated body fluid conditions. Mater Characterization. 2002;49(1):73-9. [Google Scholar]
- Patil P, Kharbanda O, Duggal R, Das T, Kalyanasundaram D. Surface deterioration and elemental composition of retrieved orthodontic miniscrews. Am J Orthod Dentofacial Orthop. 2015;147(4):S88-100. [Google Scholar]
- Abstract
- Introduction
- Materials and Methods
- Study material include
- Methodology
- L929 cell culture formation
- Cytotoxicity assays
- Determination of cell viability with the help of crystal violet (CV) colorimetric assay
- Determination of cellular metabolism with the help of MTT colorimetric assay
- Corrosion resistance
- Determination of corrosion resistance by Field emission scanning electron microscopy (FESEM)
- Determination of corrosion products by inductively coupled plasma – optical emission spectrometry (ICP-OES)
- Statistical procedures
- Results
- Discussion
- Conclusion
- Conflict of Interest
- Source of Funding
- Ethical Approval
- Acknowledgement
- References
How to Cite This Article
Vancouver
Sayyed S, Kalia A, Hegde A, Mirdehghan N, Shaikh W, Chaudhary T. A comparative evaluation of cytotoxicity and corrosion resistance between titanium and stainless steel orthodontic mini implants: An in vitro study [Internet]. Int J Oral Health Dent. 2024 [cited 2025 Oct 01];10(3):223-230. Available from: https://doi.org/10.18231/j.ijohd.2024.039
APA
Sayyed, S., Kalia, A., Hegde, A., Mirdehghan, N., Shaikh, W., Chaudhary, T. (2024). A comparative evaluation of cytotoxicity and corrosion resistance between titanium and stainless steel orthodontic mini implants: An in vitro study. Int J Oral Health Dent, 10(3), 223-230. https://doi.org/10.18231/j.ijohd.2024.039
MLA
Sayyed, Sofiya, Kalia, Ajit, Hegde, Ashwith, Mirdehghan, Nasim, Shaikh, Waheed, Chaudhary, Tahreem. "A comparative evaluation of cytotoxicity and corrosion resistance between titanium and stainless steel orthodontic mini implants: An in vitro study." Int J Oral Health Dent, vol. 10, no. 3, 2024, pp. 223-230. https://doi.org/10.18231/j.ijohd.2024.039
Chicago
Sayyed, S., Kalia, A., Hegde, A., Mirdehghan, N., Shaikh, W., Chaudhary, T.. "A comparative evaluation of cytotoxicity and corrosion resistance between titanium and stainless steel orthodontic mini implants: An in vitro study." Int J Oral Health Dent 10, no. 3 (2024): 223-230. https://doi.org/10.18231/j.ijohd.2024.039
